Why GMP Compliance Is Paramount For High-Quality Mesenchymal Stem Cell?
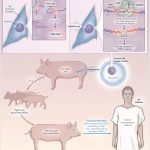
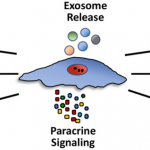
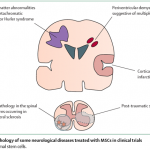
The past twenty years have witnessed a fascinating unboxing of the mesenchymal stem cells. These microscopic marvels hold the potential to revolutionize how we approach disease. These mesenchymal stem cells are extracted from adult cells, and hence, they rarely receive any ethical backlash. Unlike most stem cells, mesenchymal ones boast remarkable versatility and can morph into diverse cell types, from bone to blood vessels. These cells carry several regenerative and anti-inflammatory prowess, which has propelled them to the forefront of stem cell therapy. The use in therapeutic applications has ignited a surge in demand that outpaces our current production capabilities.
Given the surge in demand for MSCs for research, providing researchers with high-quality stem cells for reproducible research is necessary. Enter the realm of Good Manufacturing Practices (GMP), our roadmap towards building factories for these cellular powerhouses, ensuring not just quantity but unparalleled quality and safety. Buckle up, science researchers, for we’re about to delve into the intricate dance of scaling up MSC production while upholding the highest standards, paving the way for a future where these microscopic maestros weave their magic on a grand scale.